Witnessing Protocols
The option to enable counter-signing is a group policy that can be activated by the group's administrator. Navigate to My Groups and click Edit. Open the Group Policy tab to activate the witness sign option for your lab.

Check the option Witness Signature is required for all experiments and click Save. Note: as signatures are an optional setting for protocols, the Signature is required to finalize protocols setting must also be activated in order to use the witness signature feature.
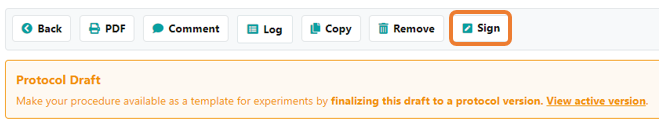
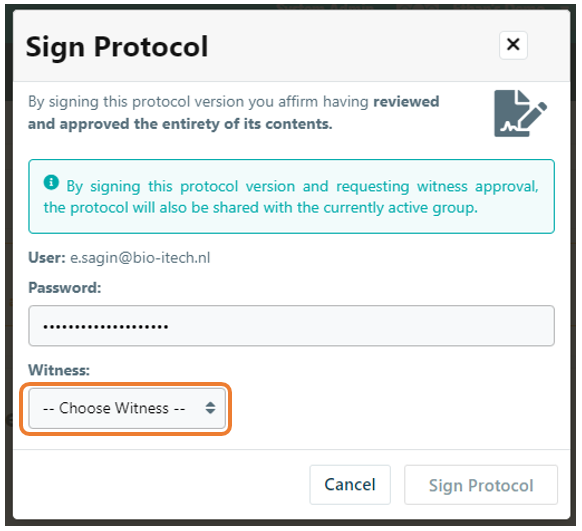
Once the witness signature option is active, protocols will require a second signature before locking. When signing a protocol, you will now have the option to assign a witness to counter-sign the protocol. Select the witness from the list of users in your lab. Note that only users with permission to witness-sign protocols will be listed. Once a protocol is signed, the protocol will be temporarily locked from further modification. Note: the witness' signature is required in order to publish the protocol. If the witness declines the signature request, then the protocol will be unlocked for further editing in draft mode.
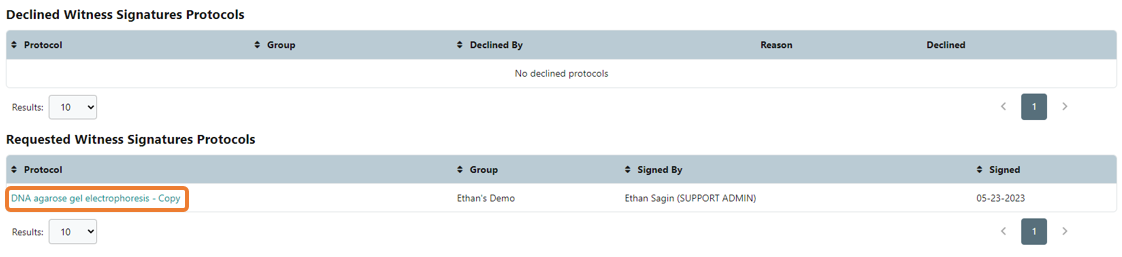
The assigned witness will receive a notification of the signature request. and the protocol will be listed on their Dashboard under the Pending Signature header. If you have installed the eLabJournal Mobile App, you will also receive a notification on your mobile device.
On the signed protocol's page, the witness has the option to approve and witness sign the experiment or to decline and unlock the protocol. In the latter case, the user that created the protocol receives a notification with the witness' note about how to adjust the protocol before resubmitting it for signing.
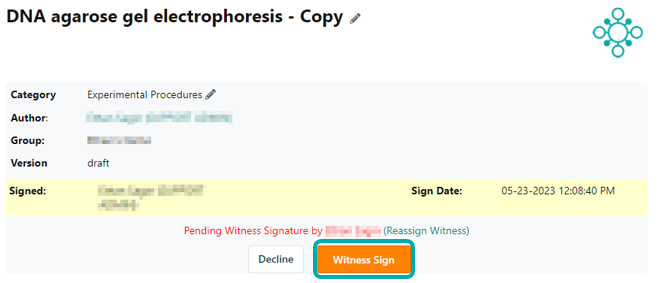
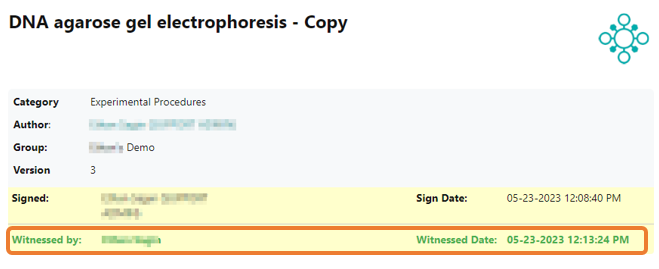
Once the protocol is counter-signed, the witness signature with timestamp is added to the protocol header. If you need to amend the signed protocol with additional information, you can open a new draft version of the protocol. If this option is insufficient, you can create a copy of the published protocol, which will automatically be linked back to the original protocol in the footer.. This approach ensures that the full audit trail of data including correct timestamps remains intact in accordance with GLP guidelines and CFR 21 part 11 regulations.